PRODUCT PORTFOLIO
We are 100% committed to addressing every segment of the aorta and advancing aortic care. We make dedicated partners, delivering on today's and tomorrow's needs, through our Endovascular, Hybrid, and Surgical platforms.
The Decision is Yours.
SURGICAL PORTFOLIO
Gelweave™ Confident Aortic Repair
Built with precision, you can trust in Gelweave for the confidence to confront the challenges of aortic repair.
Gelsoft™ Plus Feel the Difference
Designed with care, feel the difference with Gelsoft Plus, the knitted aortic repair solution for your patients.
Explore our expansive range of surgical grafts, over 40 years in the making.
PROUD TO BE PART OF THE STORY
Watch and read inspiring stories from individuals like you who have benefited from our devices. Their stories are as genuine as it gets, offering a candid look at the remarkable journeys they've been on.
VISION & MISSION
Together with our customers, we strive to create effective management of aortic disease and drive the evolution of our cardiovascular solutions for our patients.
FEATURES & BENEFITS
Crafted with adaptability and versatility as our guiding principles, our products adapt to meet the needs of your patients.
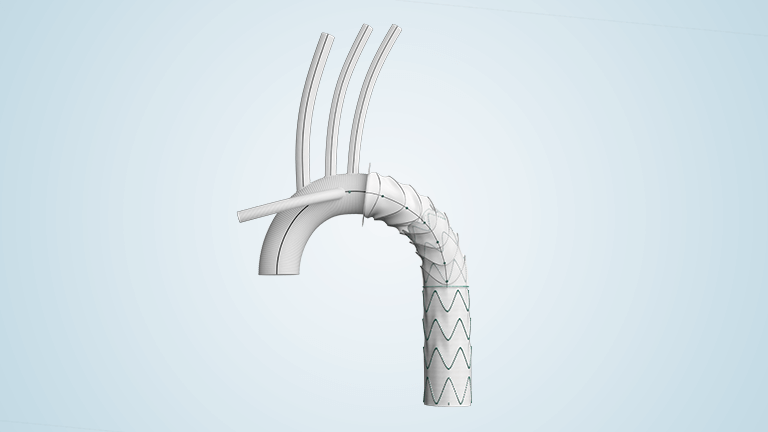
Discover products for aortic repair
Product availability subject to local regulatory approval.
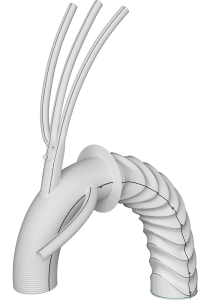
Explore Custom Solutions for aortic repair
The availability of custom-made devices is subject to local regulatory guidelines. Caution: not available in the USA.
Careers at Terumo Aortic